|
3/18/2023 0 Comments The law of entropy Therefore it makes sense that they do not specifically address the why and how of heat transport. 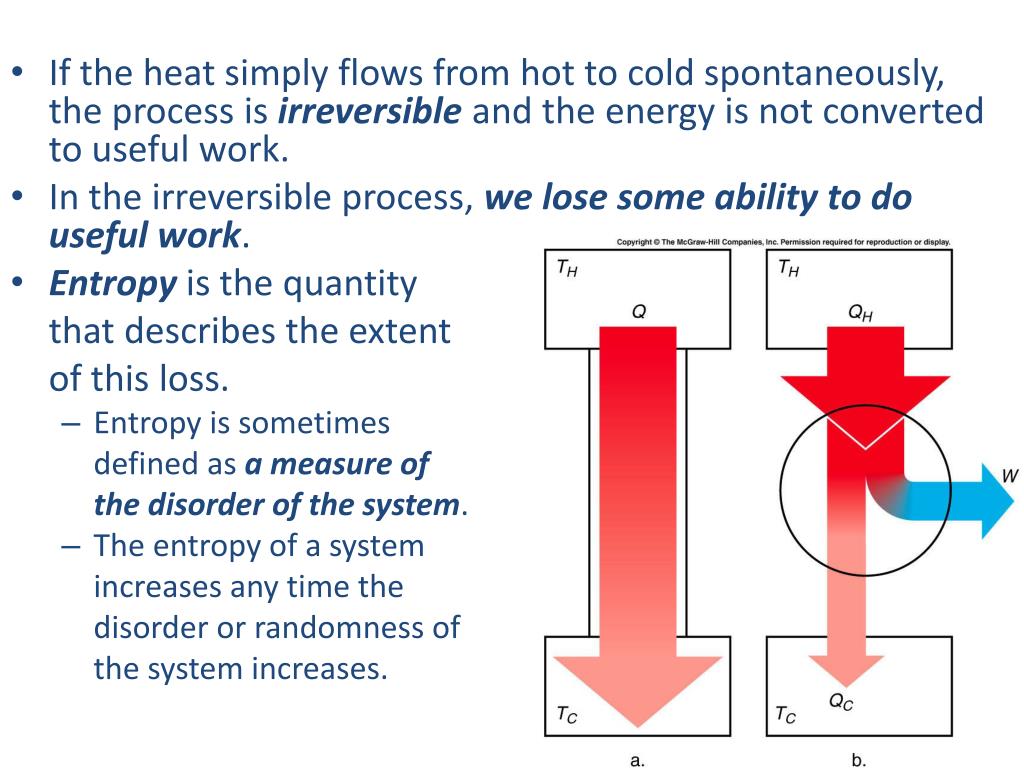
Thermodynamics laws were introduced before the full acceptance of the atomic theory. These thermodynamic principles describe how these quantities act in different situations. The law of thermodynamics elaborates on the physical quantities like radiation, temperature, and entropy governed by the thermodynamic system at thermal equilibrium. The laws of thermodynamics use a collection of basic physical quantities to describe how a thermodynamic system evolves. It indicates that it works with the system to support rather than the way matter is made up of molecules. It depends on the starting and ending conditions of the transformation. It is also known as macroscopic science and doesn’t give any significant importance to the mechanism and speed of these energy exchanges. The movement of microscopic particles inside an object produces this heat the quicker these particles travel, will produce more heat. 
The energy that heat produces is known as thermal energy. It specifically discusses how thermal energy is changed into or out of other types of energy and how this cycle affects matter. 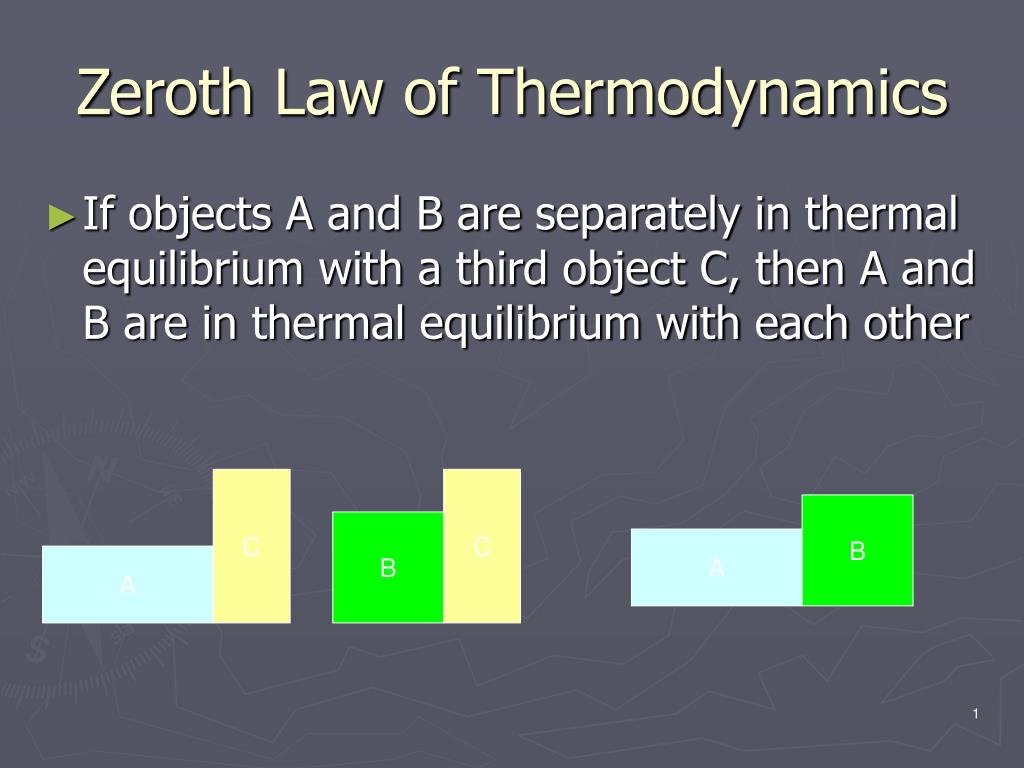
Physics’ branch of thermodynamics examines how temperature, work, and heat are related to radiation, energy, and the physicochemical properties of matter. The evolution of these values is governed by the four laws of thermodynamics, which also provide a numerical representation. The science of thermodynamics examines the ideas of temperature, heat, and the interactions between heat and other energy sources. William Thomson first used the term thermodynamics in 1749. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
